Introduction
Dialysis is a life-sustaining treatment currently keeping more than 800,000 Americans with kidney failure alive. For many patients undergoing three or more sessions each week, the procedure feels routine—yet most people understand what dialysis does without knowing how it actually works at a physical level.
Dialysis is not simply "cleaning blood" through a filter. It relies on three specific physical transport processes—diffusion, osmosis, and convection—that determine what gets removed, how fast, and how effectively.
This guide breaks down each transport process in plain language so patients, caregivers, and families can understand what is happening during every dialysis session—and why decisions about treatment timing, duration, and treatment type directly affect outcomes.
TLDR
- Dialysis replaces the filtering work of failed kidneys using three physical transport processes to move waste and excess fluid out of the blood
- Diffusion, osmosis, and convection each play a distinct role—moving waste, drawing out excess water, and using pressure to clear dissolved solutes
- A semipermeable membrane separates blood from dialysis fluid—it lets waste particles through while blocking blood cells and proteins
- Hemodialysis relies mainly on diffusion and convection; peritoneal dialysis uses diffusion and osmosis through the abdominal lining
- Dialysis effectively removes urea, creatinine, excess potassium, and water, but cannot fully replicate hormone production or all kidney functions
What Is Dialysis and Why Do Transport Processes Matter?
Dialysis is a medical treatment that mechanically performs key kidney functions when the kidneys can no longer do so. Specifically, it removes waste products, excess fluid, and harmful electrolytes from the blood. According to KDIGO 2024 Clinical Practice Guidelines, the decision to initiate dialysis should be based on symptoms, quality of life, and laboratory abnormalities rather than a strict eGFR threshold, though this typically occurs when eGFR is between 5 and 10 mL/min/1.73m².
Healthy kidneys contain approximately 1 million filtering units called nephrons that filter about 150 quarts of blood every day. Dialysis must replicate this workload using physical principles applied across an artificial or biological membrane.How well those mechanisms function determines the effectiveness of every session.
That said, dialysis has real limitations. It cannot cure kidney disease, replicate hormone production (such as erythropoietin, which signals red blood cell production), or match the precision healthy kidneys provide for long-term acid-base balance. Understanding these boundaries — and the transport processes that drive what dialysis can do — is what this guide covers.
The Three Transport Processes That Power Dialysis
Dialysis works through three distinct physical mechanisms, each moving different substances across a semipermeable membrane in different ways. Understanding each one separately makes the overall process much clearer.
Diffusion: Moving Waste Down a Concentration Gradient
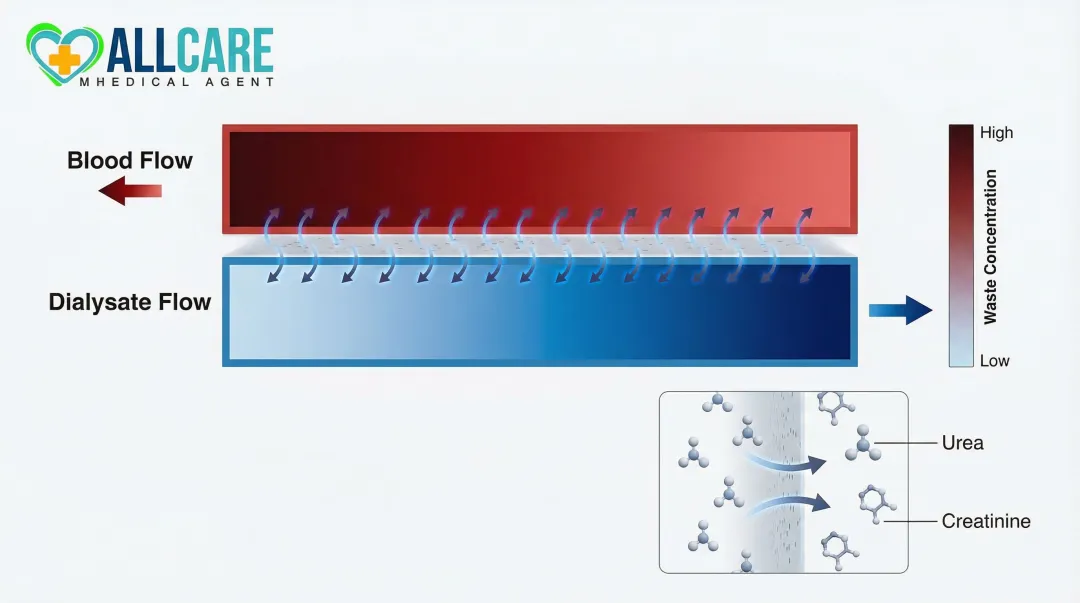
Diffusion is the movement of dissolved particles (solutes) from an area of higher concentration to an area of lower concentration across a semipermeable membrane. This is the primary mechanism for removing waste products like urea and creatinine from the blood.
A drop of dye spreading through a glass of water follows the same principle — dialysis places waste-loaded blood on one side of a membrane and fresh, low-waste dialysate fluid on the other. Waste naturally migrates across because the concentration gradient pulls it in that direction.
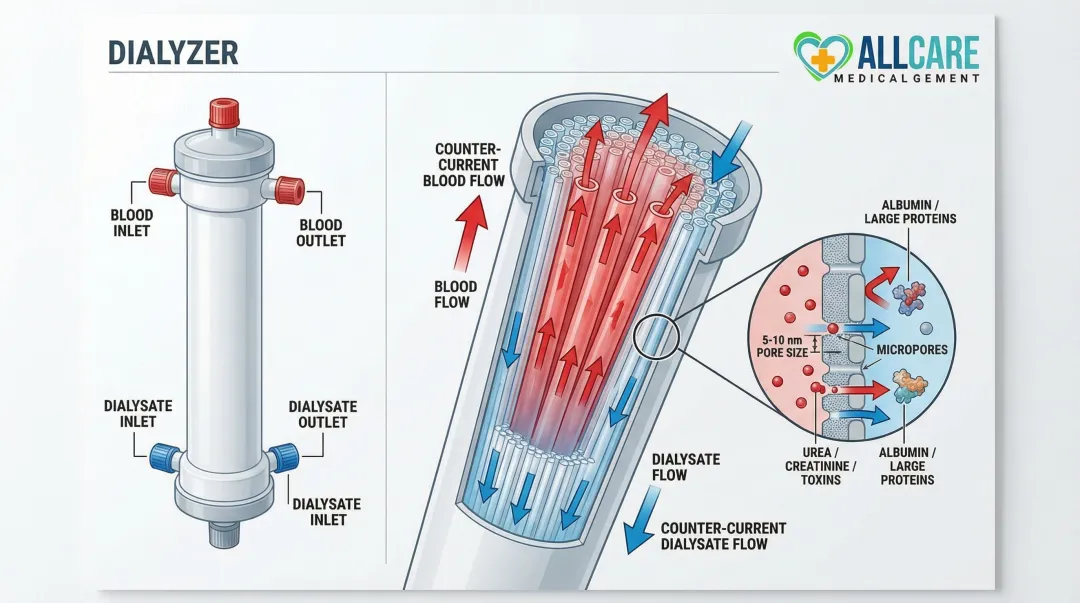
Small, water-soluble molecules such as urea and creatinine are effectively cleared this way. To maximize efficiency, modern dialyzers use counter-current flow design, where blood and dialysate flow in opposite directions. This maintains a continuous high concentration gradient across the entire membrane length, making diffusion far more efficient than parallel flow would allow.
Osmosis: Drawing Excess Fluid Out of the Body
Osmosis is the movement of water—not solutes—across a semipermeable membrane from an area of lower solute concentration to an area of higher solute concentration. In practical terms, this is how excess water is removed from the blood, particularly in peritoneal dialysis.
Dialysis fluid in peritoneal dialysis is formulated with a high concentration of glucose (dextrose) or other osmotic agents. This creates an osmotic pull that draws water out of surrounding blood vessels and into the dialysate fluid — the same way salt draws moisture out of vegetables.
For long dwells (such as overnight in continuous ambulatory peritoneal dialysis), icodextrin (brand name Extraneal) is used as a colloid osmotic agent. Icodextrin exerts osmotic pressure across small intercellular pores, improving ultrafiltration and the clearance of creatinine and urea nitrogen compared to standard dextrose solutions.
Important distinction: In hemodialysis, excess fluid removal is controlled through transmembrane pressure (a form of convection) rather than pure osmosis — which is why glucose-based osmotic agents are central to peritoneal dialysis but not hemodialysis.
Convection: Using Pressure to Push Waste Through
Convection (also called ultrafiltration) is the movement of water and dissolved particles across a membrane driven by a pressure difference rather than a concentration gradient. Pressure forces fluid through the membrane, and dissolved waste gets dragged along with it — a phenomenon known as solute drag.
Hemodialysis machines apply transmembrane pressure across the dialyzer membrane, forcing water and dissolved waste to move together into the dialysate. This is particularly effective at removing larger middle-molecule waste products — such as beta-2 microglobulin — that diffusion alone cannot efficiently clear.
Advanced approach: Hemodiafiltration (HDF) combines both diffusion and high-volume convection to improve clearance of a broader range of waste molecules. While online HDF has long been standard in Europe, it was largely unavailable in the United States until FDA granted 510(k) clearance to HDF-capable systems in 2024 and 2025, paving the way for broader U.S. adoption.
How a Dialysis Session Puts These Processes to Work
How Each Dialysis Type Gets Started
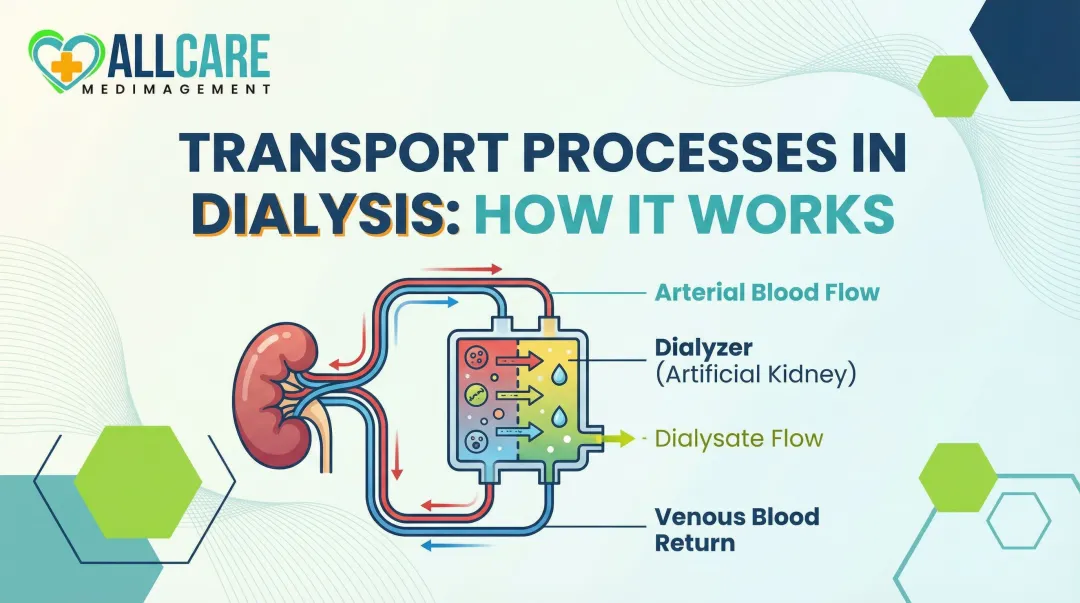
In hemodialysis, blood is drawn from a vascular access point — an arteriovenous fistula, graft, or catheter — circulated through the dialyzer at a controlled flow rate, and returned to the body. In peritoneal dialysis, dialysate is instilled through a catheter into the abdominal cavity, where it dwells against the peritoneal membrane.
Both setups channel blood and dialysate into close proximity across a semipermeable membrane. In hemodialysis, that membrane lives inside the dialyzer: a device containing thousands of hollow fibers. Blood flows through the fiber interiors while dialysate flows in the opposite direction along the outside — a countercurrent arrangement that keeps concentration gradients steep and diffusion efficient. The fiber membrane is what makes all three transport processes (diffusion, convection, and osmosis) possible.
Session Duration and Efficiency
A standard in-center hemodialysis session lasts 3–5 hours because it takes that long for blood to cycle through the dialyzer enough times to achieve adequate clearance. During this time, the patient's blood makes multiple passes through the extracorporeal circuit. Because solutes like creatinine and urea must redistribute slowly between red blood cells and plasma, multiple passes are needed to pull enough waste out of the full blood volume.
The Output Phase
As dialysate absorbs waste through diffusion and convection (or osmosis in PD), it becomes increasingly saturated with:
- Urea (nitrogen waste from protein metabolism)
- Creatinine (a muscle metabolism byproduct)
- Excess potassium, phosphorus, and sodium
- Excess water
The spent dialysate is discarded, and the now-cleaned blood is returned to the body. Whether that removal was sufficient is what adequacy testing measures.
Monitoring and Adequacy Testing
Dialysis adequacy is measured to assess whether the transport processes are working effectively enough. In thrice-weekly hemodialysis, this is measured using Kt/V, a urea-kinetic metric where 'K' represents dialyzer clearance, 't' is time, and 'V' is the urea distribution volume.
The NKF KDOQI 2015 guidelines recommend:
- Target single-pool Kt/V (spKt/V): 1.4 per session
- Minimum delivered spKt/V: 1.2
Lower delivered doses are strongly associated with increased patient morbidity and mortality. Patients should ask their care team about their Kt/V scores.
For peritoneal dialysis, the Peritoneal Equilibration Test (PET) assesses membrane transport adequacy by measuring the dialysate-to-plasma (D/P) creatinine ratio and ultrafiltration capacity.
Hemodialysis vs. Peritoneal Dialysis: Same Principles, Different Settings
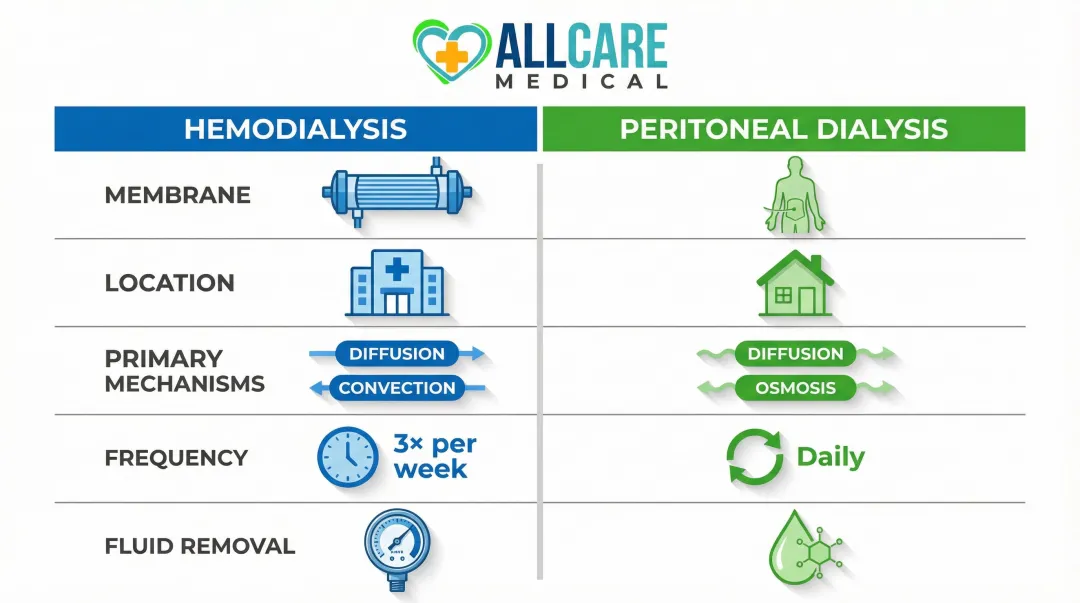
Both hemodialysis and peritoneal dialysis rely on the same underlying transport processes—diffusion, osmosis, and convection—but differ in the membrane used, the location of filtration, and the balance of mechanisms at play.
Hemodialysis
- Membrane: Artificial dialyzer with thousands of hollow fibers
- Location: Performed at a dialysis center (typically)
- Primary mechanisms: Diffusion and convection
- Frequency: Three times per week, 3–5 hours per session
- Fluid removal: Controlled by machine-applied transmembrane pressure
According to USRDS 2025 Annual Data Report, 81.7% of incident U.S. patients started in-center hemodialysis in 2023, making it the dominant modality. Because hemodialysis requires travel to a dialysis center typically three times per week, reliable transportation is a practical necessity. AllCare Medical Transport in Florida provides consistent, safe rides to and from dialysis centers for patients who need ambulatory, wheelchair, or stretcher transport.
Peritoneal Dialysis
- Membrane: The patient's own peritoneal lining
- Location: Performed at home or during sleep
- Primary mechanisms: Diffusion and osmosis
- Frequency: Continuous or through scheduled daily exchanges
- Fluid removal: Osmotic gradient created by glucose or icodextrin in dialysate
Peritoneal dialysis reached an all-time high of 14.1% of incident patients in 2023. Because it is done at home continuously or via an automated cycler at night (automated peritoneal dialysis/APD), the transport processes operate more gradually over more hours. This is generally gentler on fluid balance, though it requires patient or caregiver training and consistent daily effort.
Key Practical Differences
| Factor | Hemodialysis | Peritoneal Dialysis |
|---|---|---|
| Clearance speed | Faster per session; machine-controlled | Gradual but continuous |
| Residual kidney function | Less protective long-term | May better preserve early-stage function (with low-GDP solutions) |
| Patient burden | Clinic travel 3×/week | Daily exchanges; home training required |
| Lifestyle fit | Structured schedule, external support | More flexible; suited to independent patients |
The best fit depends on medical status, lifestyle, and available support—decisions made with a nephrologist, not a checklist.
What Dialysis Can (and Cannot) Remove
What Dialysis Effectively Removes
Dialysis successfully removes small-molecule waste products that healthy kidneys would normally excrete through urine:
- Urea — nitrogen waste from protein metabolism
- Creatinine — a muscle metabolism byproduct; dialysis removes creatinine primarily through diffusion, since creatinine is a small-molecule solute that moves readily across the semipermeable membrane
- Excess potassium — important for preventing dangerous heart rhythm problems
- Excess phosphorus — helps prevent bone disease
- Excess sodium — important for blood pressure control
- Excess water — prevents fluid overload and heart strain
What Dialysis Cannot Fully Replace
Dialysis cannot replicate all kidney functions. Three gaps are clinically significant for long-term patients.
Hormone production: Failing kidneys do not produce adequate erythropoietin (EPO), a hormone that stimulates red blood cell production in bone marrow. Dialysis patients commonly develop anemia and require erythropoiesis-stimulating agents (ESAs) like epoetin alfa or darbepoetin alfa.
Vitamin D activation: Dialysis cannot perform the 1-alpha hydroxylation required to activate vitamin D. Patients with severe hyperparathyroidism often require active vitamin D analogs such as calcitriol.
Protein-bound toxins: Standard diffusion-based hemodialysis struggles to clear protein-bound uremic toxins, such as indoxyl sulfate and p-cresol. Because these toxins bind tightly to plasma albumin, only the small unbound fraction crosses the membrane — driving accumulation and contributing to uremic toxicity and cardiovascular risk.

For patients with high uremic toxin burden, this limitation is one reason hemodiafiltration (HDF) increasingly appears in treatment protocols.
Conclusion
Dialysis is not a black-box procedure. It operates on well-understood physical principles—diffusion, osmosis, and convection—applied through a semipermeable membrane. Knowing how these processes work helps patients ask better questions, understand why session duration and frequency matter, and recognize what "adequate dialysis" actually means.
The science runs the session, but logistics determine whether patients get the full benefit of every treatment. Consistent attendance, proper nutrition, and reliable transportation to each appointment all directly affect outcomes — missing sessions disrupts the clearance schedule that diffusion and convection depend on.
Patients and families who understand these processes are better equipped to make informed decisions about their care. If getting to and from the dialysis center is a barrier, dedicated medical transportation services — like those specializing in dialysis runs — can help protect that consistency.
Frequently Asked Questions
What are the general transport processes involved in dialysis?
Dialysis uses three transport processes: diffusion (waste moves down a concentration gradient into the dialysate), osmosis (excess water is drawn out by osmotic agents), and convection (pressure forces water and dissolved solutes through the membrane). Together, these mechanisms remove waste and excess fluid from the blood.
Does dialysis remove creatinine?
Yes, dialysis removes creatinine—a waste product of normal muscle metabolism—primarily through diffusion. Creatinine is a small-molecule solute that moves readily across the semipermeable membrane from higher concentration in the blood to lower concentration in the dialysate.
What is a semipermeable membrane and why does it matter in dialysis?
A semipermeable membrane is a thin barrier with microscopic pores that allow small waste molecules and water to pass through while blocking larger particles like red blood cells and proteins. It is the foundation that makes all three transport processes possible and determines what gets removed from the blood.
How does diffusion differ from convection in dialysis?
Diffusion moves solutes based on concentration differences with no pressure needed, while convection uses applied pressure to push water and any dissolved substances through the membrane together. Both are used in hemodialysis: diffusion handles most small-molecule waste removal, while convection handles fluid removal and some larger molecules.
Can dialysis fully replace kidney function?
Dialysis effectively replaces the kidneys' waste-filtering and fluid-removal functions, but it cannot replicate hormone production (erythropoietin) or vitamin D activation. Dialysis patients typically take additional medications to compensate for these missing functions.
How long does a typical hemodialysis session last?
A standard in-center hemodialysis session lasts 3–5 hours, performed three times per week. Session length is set by how much time the transport processes need to adequately clear waste; shorter or less frequent sessions risk inadequate clearance.